
Fig. 1. Lake Cheston, a small, 7-acre man-made lake atop the Cumberland Plateau in Sewanee, TN. Dr. Martin Knoll’s hydrology class seen collecting data from canoes. Photo by Clark Lupton.
There is only one natural lake in all of Tennessee. Reelfoot Lake in northwest Tennessee was formed by ground subsidence during a series of great earthquakes in 1811/12. All other lakes in the state, including the many dozens on the Cumberland Plateau, are man-made. A quick look at a topographic map or Google Earth image reveals the origins of these lakes. The lakes all have a long, straight edge along part of their shore – a dam (Fig. 2). Lakes on the Plateau tend to be small, especially compared to larger impoundments on the Tennessee River and its tributaries. Lake Cheston in Sewanee, Tennessee is a good example of a typical lake on the Plateau (Figs. 1 & 2).

Fig. 2. A topographic map of a portion of Sewanee, TN. Notice the dams on the southern end of Lake Cheston and the northwestern side of the smaller Farm Pond.
Even the smallest of Plateau lakes show seasonal fluctuations in chemical and physical properties seen in larger lakes across the temperate zones of the world. As air temperature and solar radiation change through the seasons, lakes go through annual cycles of stratification and mixing.
Summer Stratification
During the summer months the maximum amount of solar radiation beams down upon a lake, warming its waters to a depth of several feet. The summer air above the lake also warms the upper lake portion. Wind blowing across the lake surface mixes the upper lake so that there is a relatively uniform temperature found to a depth of about 6 to 8ft. This layer is referred to as the epilimnion (Fig. 3).
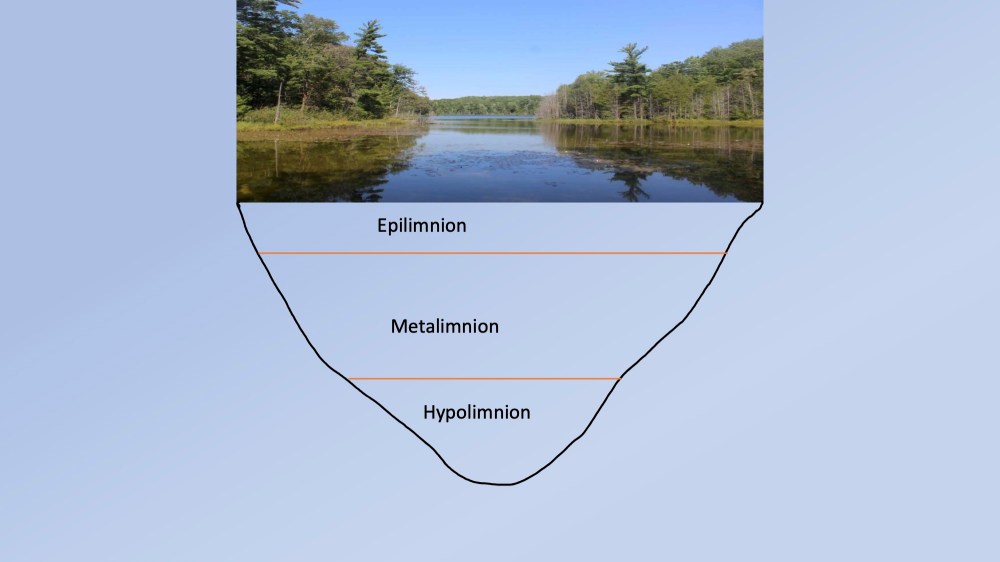
Fig. 3. The three layers found within a temperate lake during the summer months. The layers can be best detected by trends in temperature and dissolved oxygen with depth.
The density of water increases as it gets colder, until a temperature of 4 degrees C is reached. At this point water begins to expand again, becoming less dense. For a lake in summer, this means that the warmest, least dense water will be near the surface (epilimnion) while the coldest, densest water will be at the bottom. This bottom layer is uniformly cold and has no currents to mix it. It is called the hypolimnion. Below the epilimnion and out of reach of the warming effects of the sun and air, the temperature rapidly decreases with increasing depth. This zone is called the metalimnion. Swimmers that drive their toes below a depth of 6-8 ft will feel the distinct cooling within the upper parts of the metalimnion (Figs. 3 and 4).

Fig. 4. Temperature-depth profiles for Lake Cheston in the summer (pink line) and winter. Summer stratification is evident, while the winter profile shows destruction of stratification by fall turnover.
Because the epilimnion is well mixed by wind, oxygen from the atmosphere has also been dissolved throughout this uppermost lake level. In fact, the epilimnion can often be detected by uniformly high levels of dissolved oxygen (DO) in the upper 6-8 ft of the lake (Fig. 5).
Along with summer stratification by temperature and DO, there is also stratification based on chemistry in many Plateau lakes. The upper rock layers of the Plateau upon which the lakes are built are made up of sandstones, conglomerates and shales. These contain high amounts of iron (Fe)- and manganese (Mn)- bearing minerals (see previous post titled “Why we’ve pulled the plug on well water on the Cumberland Plateau”). These elements are continuously released by weathering and enter lake waters. In the summer the waters beneath the epilimnion contain little to no DO (Fig. 5). This means that the Fe and Mn can remain suspended within this low-oxygen layer. In the epilimnion there is enough DO to cause the Fe and Mn to oxidize, form a solid and settle to the lake floor. Summer concentrations of Fe and Mn in the hypolimnion typically exceed 10,000 parts per billion (ppb) and 1500 ppb, respectively. In contrast, surface concentrations are normally near 150 ppb Fe and <10 ppb Mn (Fig. 6).

Fig. 5. Graph showing relationship between dissolved oxygen and depth for summer and winter in Lake Cheston. The epilimnion can clearly be seen in the summer profile. ppm = parts per million.
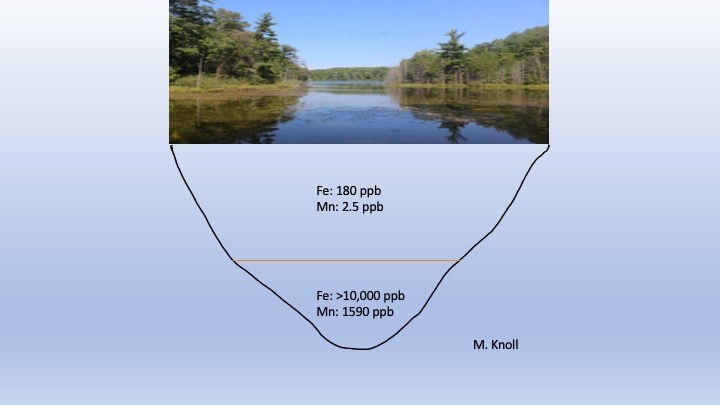
Fig. 6. Diagram showing summer stratification by chemistry in Lake Cheston. The low DO levels in the hypolimnion lead to highly elevated levels of suspended Fe and Mn (ppb = parts per billion).
Most lakes have some leakage of bottom waters from the base of their dams. As this oxygen-deprived water is exposed to the atmosphere upon leaving the dam base, the Fe and Mn oxidize through the aid of Fe- and Mn – depositing bacteria and precipitate as a red solid on stream floors (Fig. 7). In the early 2000s, the Tennessee Department of Environment and Conservation (TDEC) became concerned that leakage from Plateau dams would bring high levels of the metals far downstream into watersheds, compromising water quality there. Thus they instituted a temporary moratorium on Plateau lake construction. Research I conducted with students showed, however, that stream Fe and Mn concentrations reached normal levels at around 3000 ft downstream of dams.

Fig. 7. Fe and Mn precipitated by bacteria at the base of Brushy Lake dam in Sewanee, TN. Levels of these metals typically return to normal around 3,000 ft downstream of dams.
Some lakes are very shallow and therefore don’t have the range of temperatures to support a fully stratified water body in summer. Brushy Lake in Sewanee is only 8 ft deep and therefore has warm water from surface to base (an epilimnion) (Figs. 8 & 9).

Fig. 8. Brushy Lake, an 8 ft deep impoundment in Sewanee, TN.
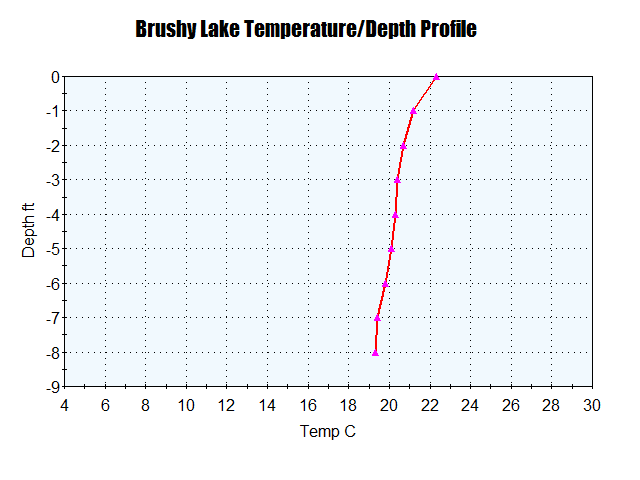
Fig. 9. Summer Temperature/Depth profile for Brushy Lake, showing an epilimnion that extends to the lake floor.
Fall Turnover
During the fall the drop in air temperature and decrease in solar radiation lead to a cooling of the uppermost lake layer. This in turn means that surface waters become more dense than the waters at greater depth. This instability, with the aid of fall winds pushing surface waters to one side of the lake, results in the upper layer of water dropping to the bottom of the lake in what is termed fall turnover. This mixing of the lake destroys the summer stratification and the lake takes on its typical winter profile, with relatively constant temperatures from surface to bottom (Fig. 4).

Fig. 10. Graph showing relationship between air temperature and surface water temperature in Lake Cheston through the fall.
The diagram above (Fig. 10) shows how decreasing air temperature in the fall causes a decrease in surface water temperature of Lake Cheston, culminating in fall turnover sometime around November 17th. After turnover the lake has a uniform distribution of dissolved oxygen (DO) from surface to bottom (Fig. 5). Notice in the dissolved oxygen graph that winter values of DO are higher overall than in summer. This is because cold water can hold more DO than warm water. Chemical stratification is also destroyed by turnover, with more or less equal values of Fe/Mn throughout the vertical extent of the lake during winter.
Another interesting pattern seen in Plateau-top lakes is zoning by pH. In Sewanee’s Lake Dimmick (also called Day Lake) in 2004, seniors in the Department of Forestry and Geology collected extensive pH data in the surface waters of the lake (Fig. 11).

Fig. 11. Lake Dimmick (Day Lake) in Sewane, TN, showing a “bathtub ring” of low pH values around its edge.
Notice that the lowest pH values form a “bathtub ring” around the edge of the lake. Rain water in the region typically has pH values between 4.5 and 5. The same values are also found in the soils that cover the Plateau. Thus precipitation falling in the lake’s watershed makes its way into the lake beneath the ground surface as groundwater flow all along the lake shore, with the original pH values essentially unchanged.
It is interesting to compare Plateau lakes to other lakes in the region. For example, is summer stratification similar in the large lakes of the Tennessee River? Below is a graph showing August values of temperature and DO near the dam in Chickamauga Lake near Chattanooga, Tennessee.

Fig. 12. Temperature and DO trends in Chickamauga Lake in early August.
Notice that the lake lacks the typical summer stratification of temperate zone lakes, with relatively uniform temperature and DO values to a depth of 56 ft. This is because the Tennessee River still flows through its impoundments. Stream flow is turbulent and results in mixing of waters. Thus, while Chickamauga Lake has the appearance of a large lake, the system is still a river with significant mixing.